Is Ccl4 Ionic Or Covalent

CCl4 (Carbon tetrachloride) is a covalent (nonpolar covalent) compound because when ane nonmetal combines with another nonmetal, information technology usually forms a covalent compound. Here, C is a nonmetal and Cl is also a nonmetal. So when they combine, it forms a covalent compound.
Well, now you lot have got to know that CCl4 is a covalent compound, simply allow me explain the in-depth reason why CCl4 is a covalent compound.
Why is CCl4 a Covalent compound?
As mentioned in a higher place, you lot can simply call back that when the nonmetal combines with another nonmetal, the bond between them is a covalent bail.
Here in CCl4, the C atom is a nonmetal and the Cl cantlet is also a nonmetal.
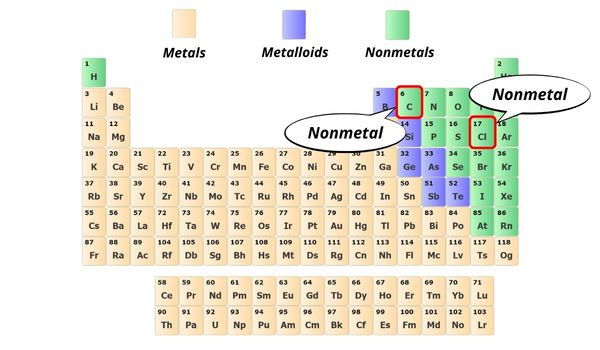
Hence the bond betwixt them is a covalent bond.
How does the covalent bond form in CCl4?
In CCl4, there are two atoms;
C and Cl.
About Carbon (C):
Carbon atom take half dozen electrons.
The electrons organisation in Carbon (C) is 2, 4.
Then the outermost orbit of Carbon cantlet has four electrons.
Now in order to achieve a stable octet, the Carbon atom needs 4 more electrons.
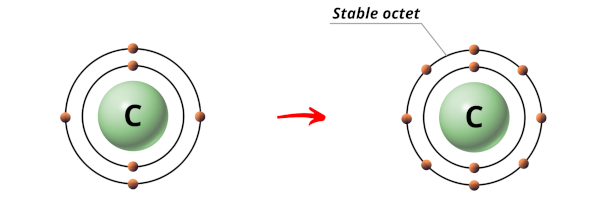
Hence during the chemical reaction, the Carbon atom will gain 4 electrons from the combining atom to form a stable octet.
Well-nigh Chlorine (Cl):
Chlorine atom accept 17 electrons.
The electrons arrangement in Chlorine (Cl) is 2, 8, 7.
So the outermost orbit of Chlorine atom has 7 electrons.
Now in order to attain a stable octet, the Chlorine atom needs 1 more electron.
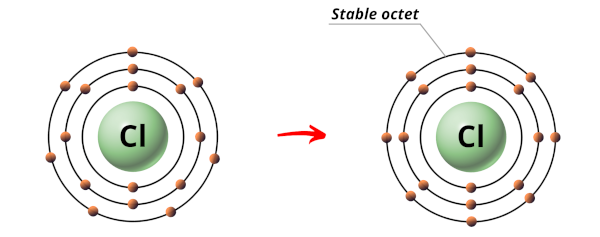
Hence during the chemical reaction, the Chlorine atom will gain one electron from the combining cantlet to form a stable octet.
What happens when C and Cl combine?
When C and Cl combine with each other, the Carbon atom and Chlorine atoms mutually share their 1-1 electrons with each other.
Because of this the carbon atom will have viii electrons in its outermost orbit and similarly the chlorine atoms will also accept 8 electrons in its outermost orbit.
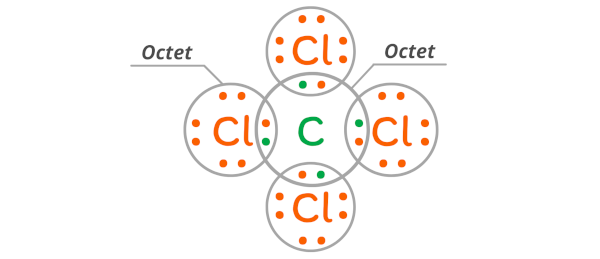
As a result, the carbon atom and chlorine atoms will take a stable octet.
And finally, equally the bail formed between the carbon and chlorine is due to the mutual sharing of electrons, it is considered a covalent bond.
Thus, CCl4 is a covalent compound.
Is CCl4 polar covalent or nonpolar covalent?
In guild to know whether CCl4 is a polar covalent molecule or nonpolar covalent molecule, nosotros have to bank check the electronegativity difference of the combining atoms.
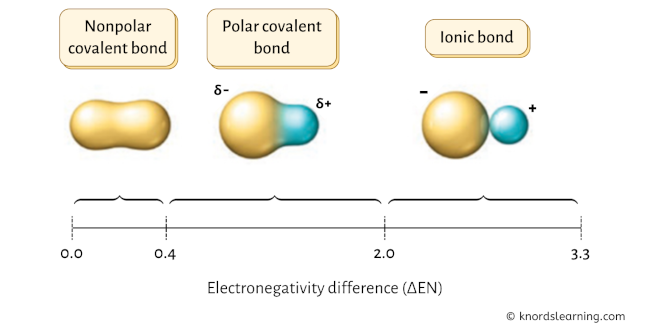
If the electronegativity difference ( ΔEN ) is less than 0.4, then the bail is nonpolar covalent bond.
If the electronegativity difference ( ΔEN ) is between 0.4 to 2.0, and so the bail is polar covalent bond.
At present the electronegativity of Carbon and Chlorine are mentioned below.
- Electronegativity of Carbon (C) = 2.55
- Electronegativity of Chlorine (Cl) = iii.16
And then for CCl4, the electronegativity difference ( ΔEN ) = iii.xvi – 2.55 = 0.61
This value lies between 0.iv to 2.0, which indicates that the bail betwixt Carbon (C) and Chlorine (Cl) is polar covalent bond.
But if you look at the 3D construction of CCl4, you can see that the structure of CCl4 is symmetrical.
Equally both the bonds (C-Cl) are symmetrical and the CCl4 molecule has a symmetrical geometry, their bail polarity gets canceled with each other.
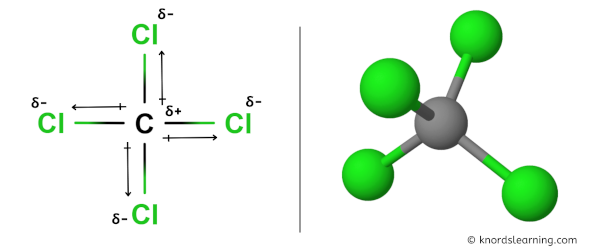
Hence, CCl4 is a nonpolar covalent molecule.
I hope you have understood the reason why CCl4 is a nonpolar covalent compound.
Check out other compounds to encounter whether they are ionic or covalent;
Is CaO Ionic or Covalent?
Is KCl (Potassium chloride) Ionic or Covalent?
Is O2 Ionic or Covalent?
Is H2S Ionic or Covalent?
Is NaBr Ionic or Covalent?
Is Ccl4 Ionic Or Covalent,
Source: https://knordslearning.com/is-ccl4-carbon-tetrachloride-ionic-or-covalent/
Posted by: wrightlizabilings.blogspot.com


0 Response to "Is Ccl4 Ionic Or Covalent"
Post a Comment